ViaCyte on Twitter: "Nov. 6: ViaCyte VP, Research and CSO Kevin D'Amour to speak at @ISSCR's “From Stem Cell Biology to New Therapies” https://t.co/f8anGrT6wi #ISSCR #biotech #Toronto #health #T1D #type1diabetes #diabetes #regenerativemedicine #

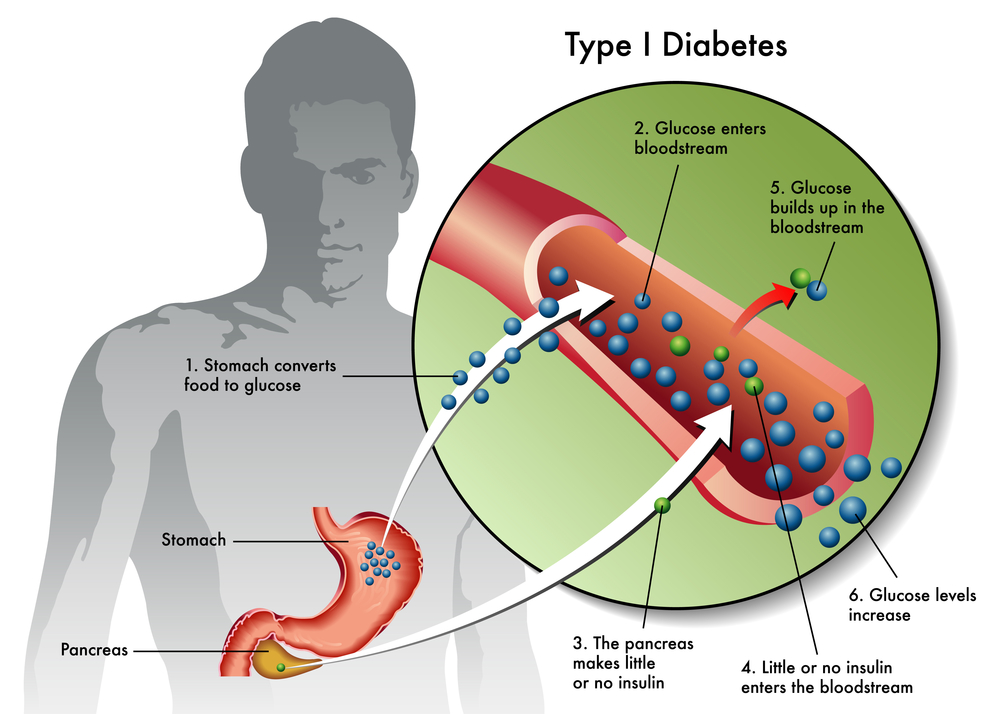
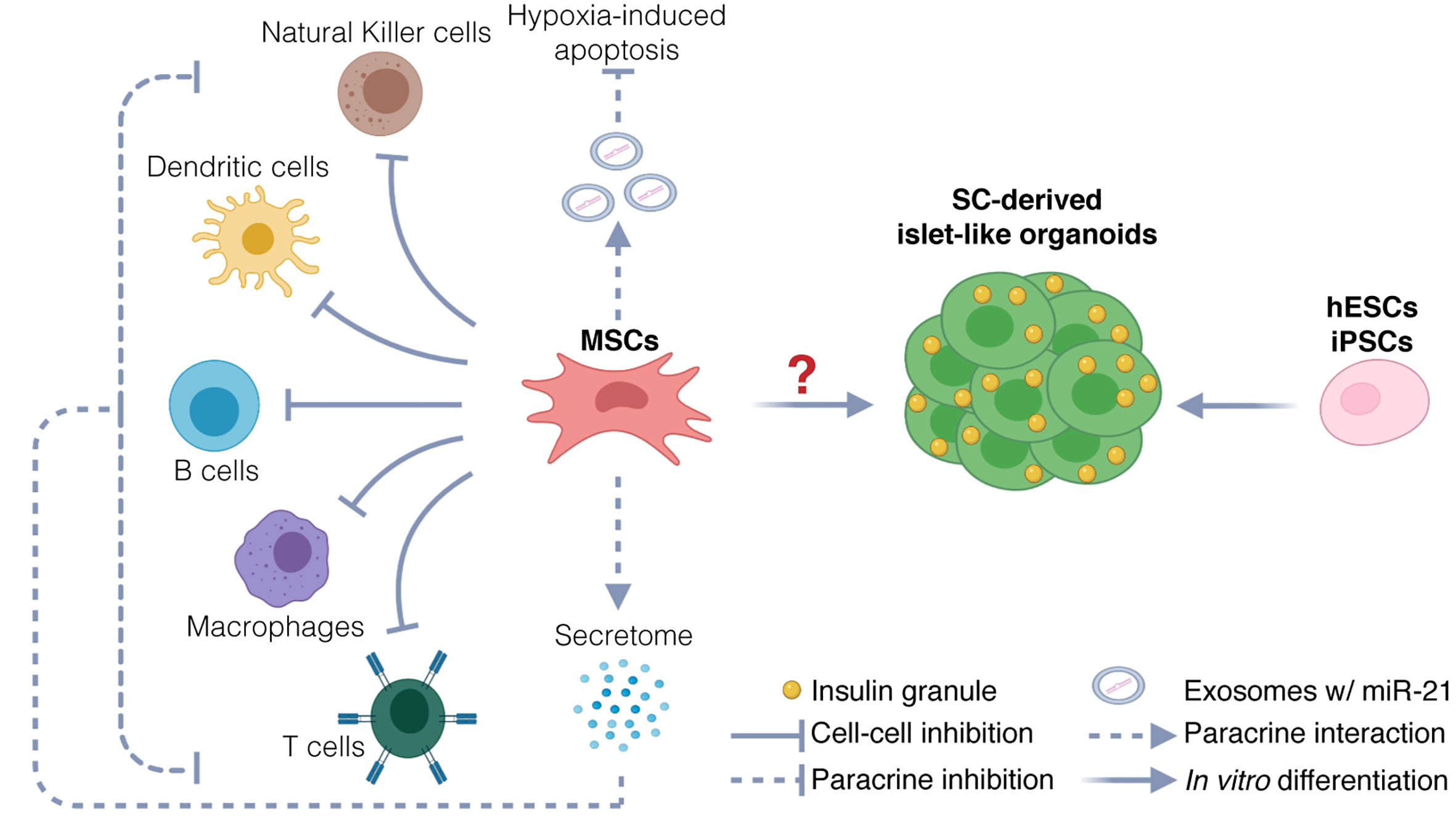
Stem cell therapies for Type 1 diabetes: current status and proposed road map to guide successful clinical trials - Senior - 2019 - Diabetic Medicine - Wiley Online Library

Stem cell therapies for Type 1 diabetes: current status and proposed road map to guide successful clinical trials - Senior - 2019 - Diabetic Medicine - Wiley Online Library

JDRF Partner ViaCyte to Immediately Initiate Type 1 Diabetes Clinical Trial — Juicebox Podcast and Arden's Day

CRISPR Therapeutics and ViaCyte, Inc. Announce First Patient Dosed in Phase 1 Clinical Trial of Novel Gene-Edited Cell Replacement Therapy for Treatment of Type 1 Diabetes (T1D)