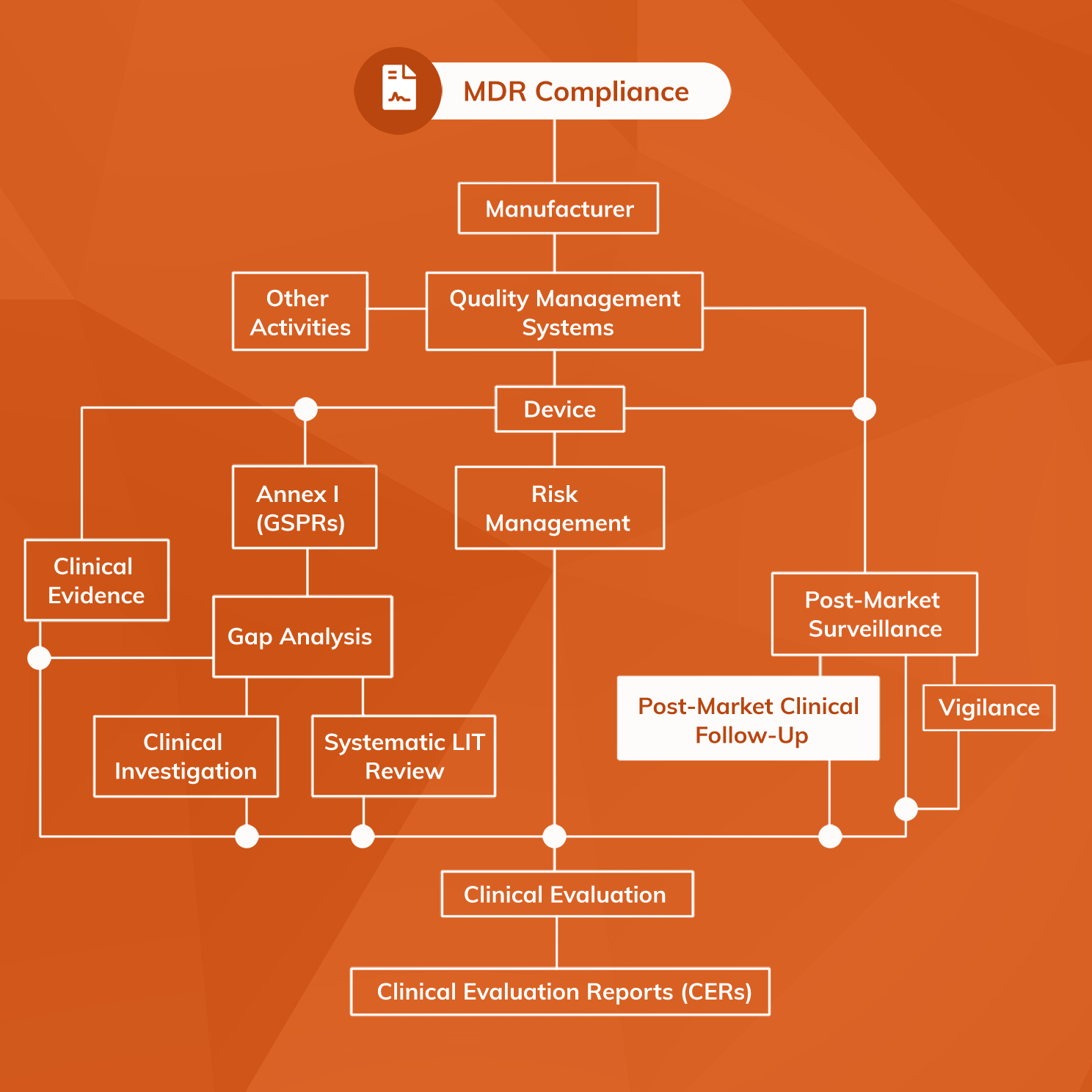
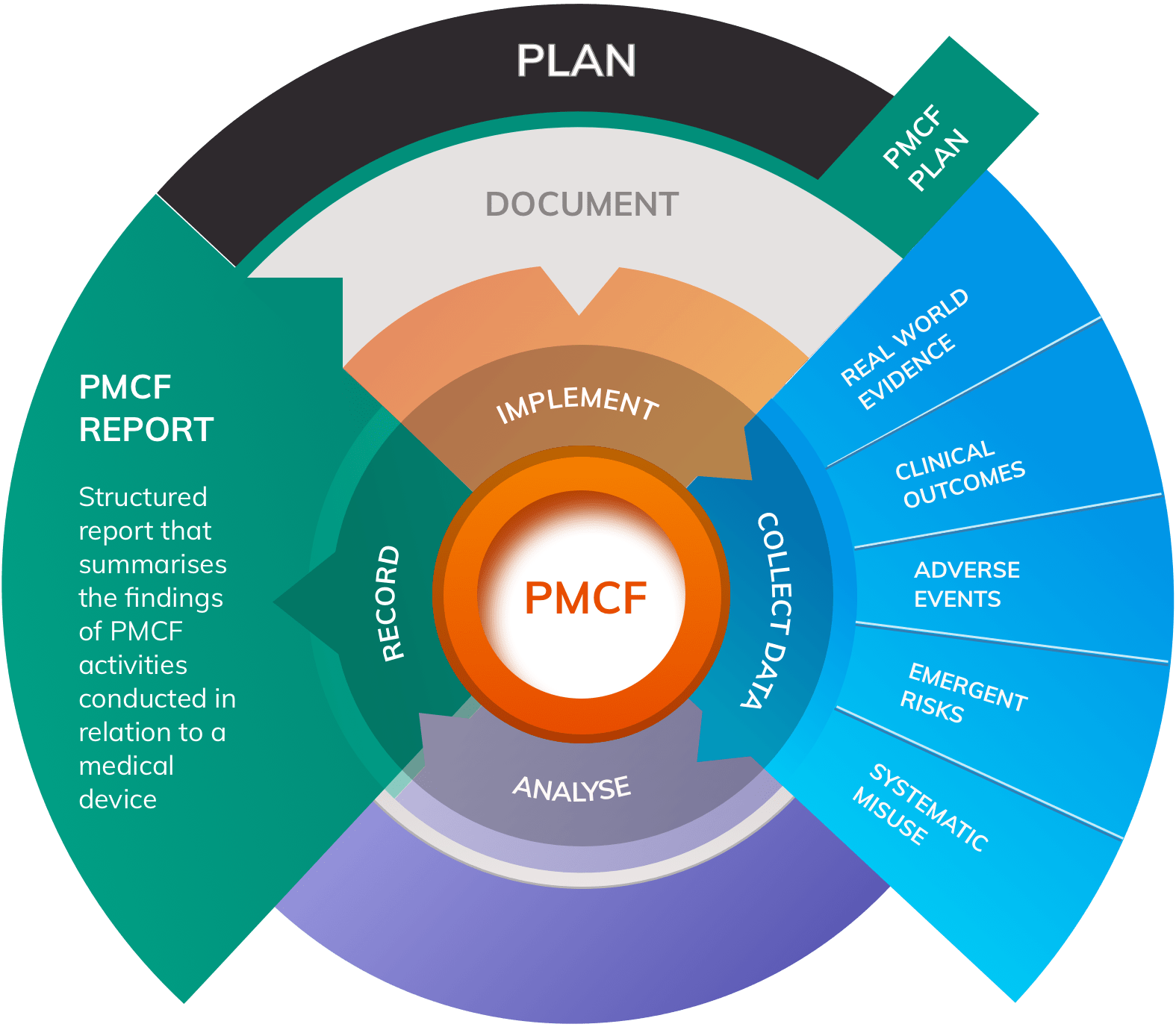
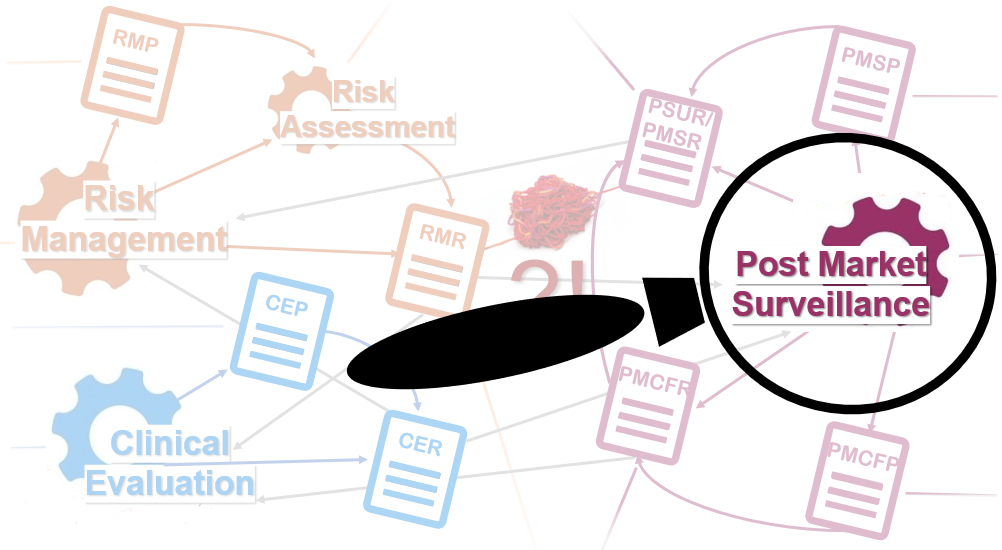
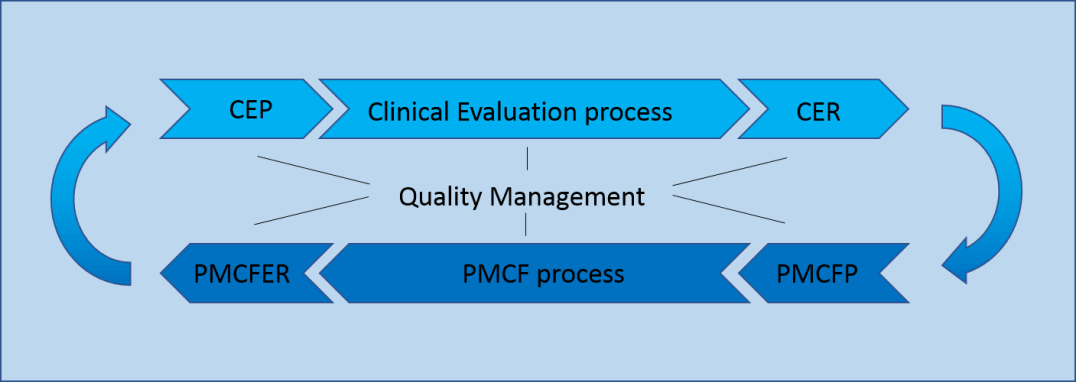
Does Your Organization's Post-Market Clinical Follow-Up (PMCF) Plan Adequately Reflect the Intensity Required in the Clinical Evaluation Report (CER) Under the Newest Medical Device Regulations? - Criterion Edge

Overview of patient enrollment, clinical follow-up and personalized... | Download Scientific Diagram