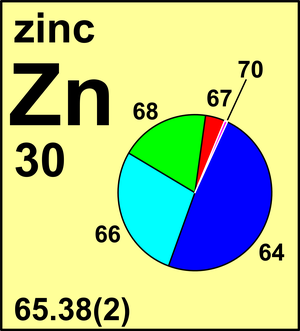
SOLVED:A 7.36-g sample of copper is contaminated with an additional 0.51 g of zinc. Suppose an atomic mass measurement is performed on this sample. What would be the apparent measured atomic mass?

Stoichiometry Chapter 11 & 12. I. Things you should remember From the Moles Unit: Identify particles as atoms, molecules (mc), and formula units (fun) - ppt download

SOLVED:CHAPTER 2 REVIEW WORKSHEET AND KEY The Mole 1) How many zinc (Zn) atoms are contained in 5.16 moles of Zn? How many moles of He are 221,000 He atoms? How many

0.277 mol ammonium acetate Convert to grams: mol NH 4 C 2 H 3 O 2 Step 1: Write the formula NH 4 C 2 H 3 O 2 Step 2: Write the given information. - ppt download

1 General Chemistry CHEM 110 Dr. Nuha Wazzan Chapter 3 Mass Relationships in Chemical Reactions. - ppt download

Section 2.5 Worksheet - class practice assignment - Atomic Masses Learning Objectives Define - StuDocu