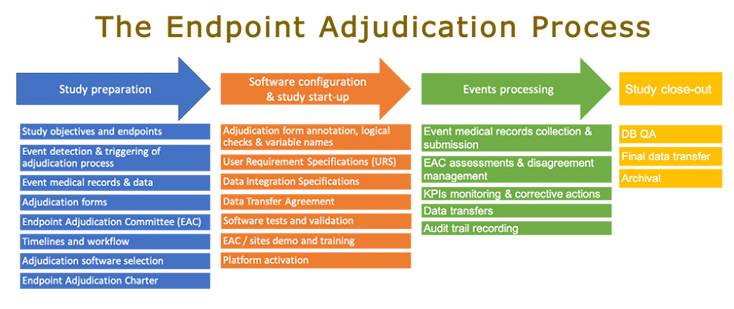
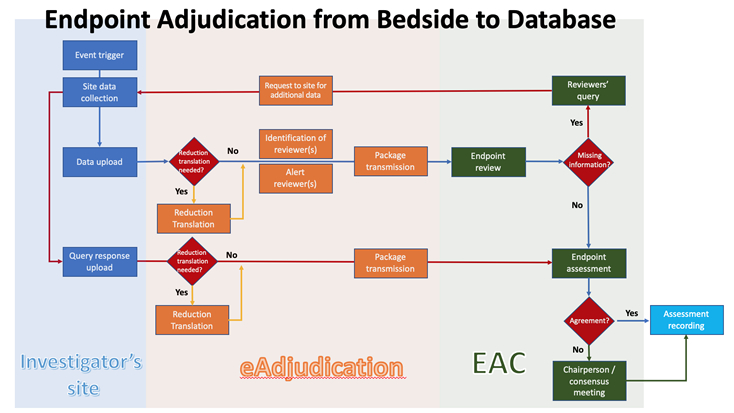
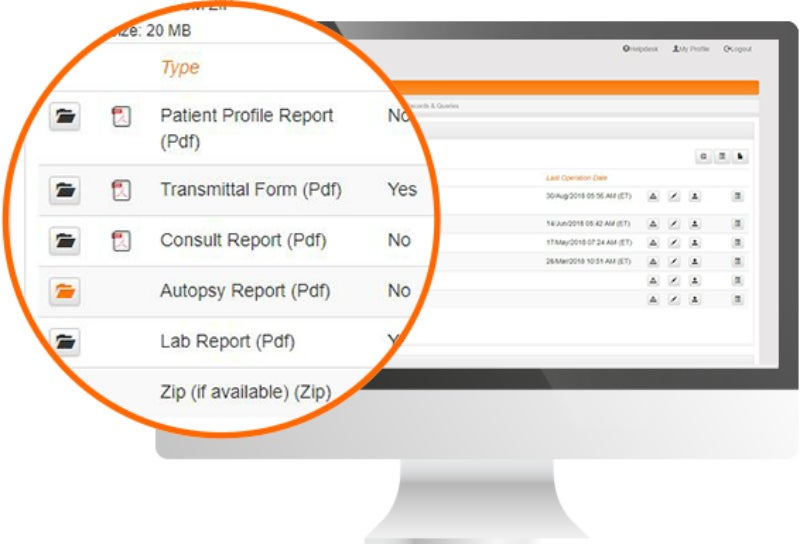
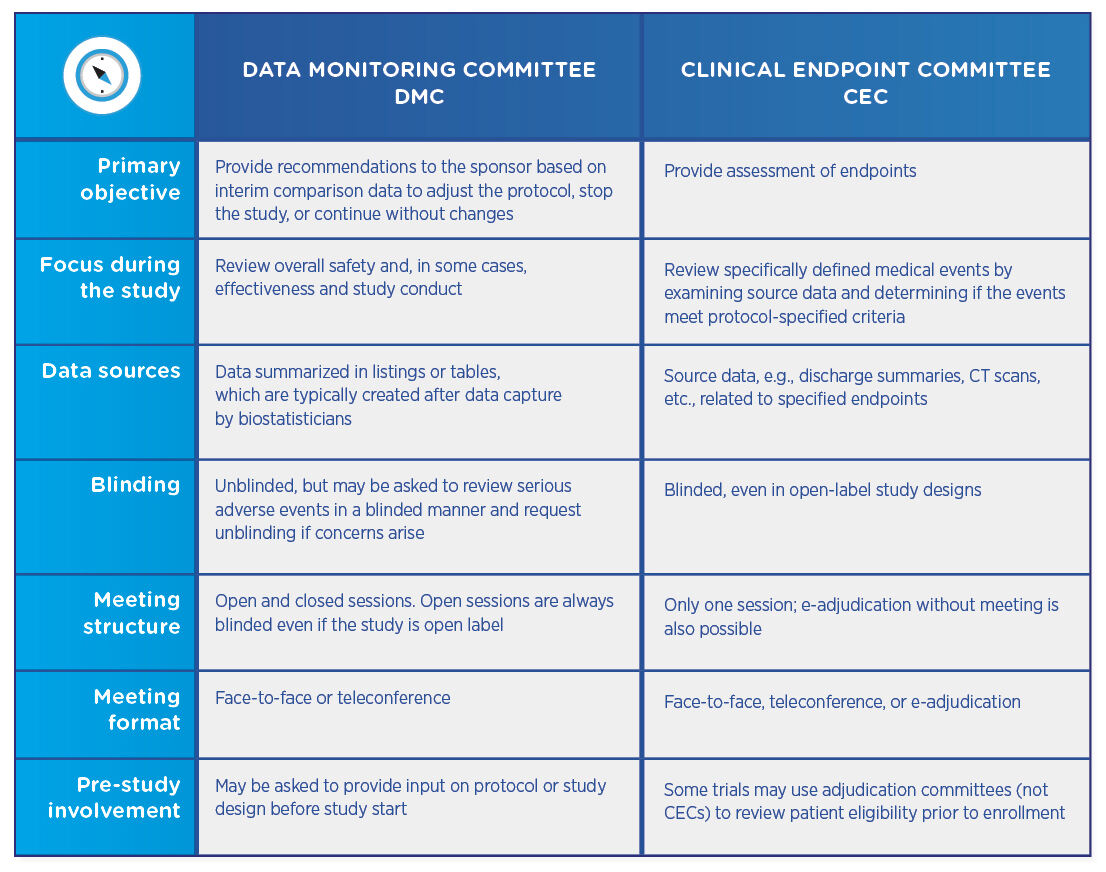
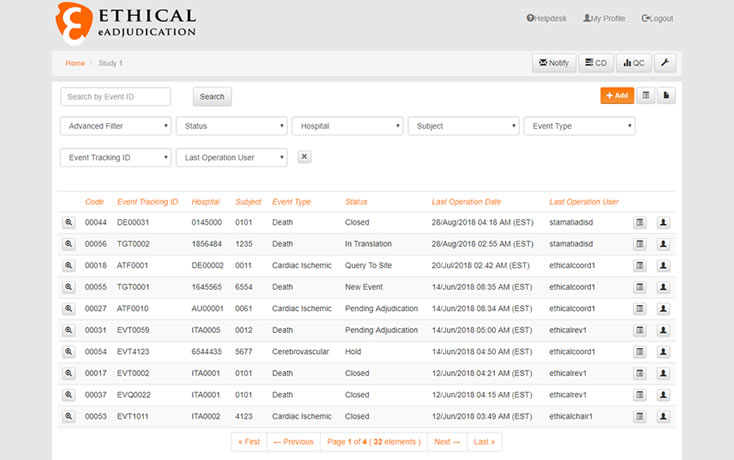
Biopharmaceutical and medical device companies have made EDC (electronic data capture) a cornerstone of clinical trial and registry management because of its proven, significant process improvements over paper-based systems. EDC is a technology ...

Comparative Reductions in Investigator-Reported and Adjudicated Ischemic Events in REDUCE-IT - ScienceDirect

Cardiovascular adverse events in the drug‐development program of bupropion for smoking cessation: A systematic retrospective adjudication effort - Kittle - 2017 - Clinical Cardiology - Wiley Online Library

PDF) Effects of the Endpoint Adjudication Process on the Results of a Randomised Controlled Trial: The ADVANCE Trial
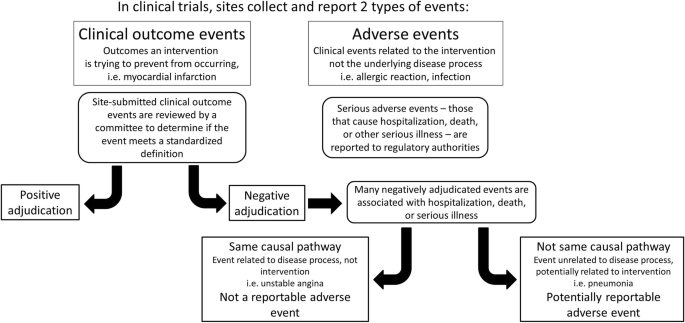
Methods for safety and endpoint ascertainment: identification of adverse events through scrutiny of negatively adjudicated events | Trials | Full Text

Agreement between public register and adjudication committee outcome in a cardiovascular randomized clinical trial Erik Kjøller, MD, DMSc, Jørgen Hilden, - ppt download
GLASSY design. CEC, Clinical Event Committee; GLASSY, GLOBAL LEADERS... | Download Scientific Diagram
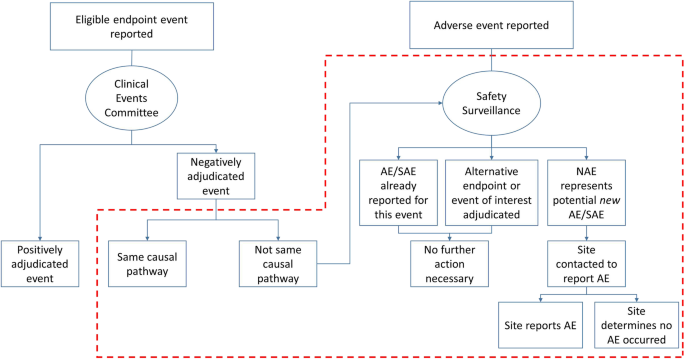
Methods for safety and endpoint ascertainment: identification of adverse events through scrutiny of negatively adjudicated events | Trials | Full Text

Central Adjudication Identified Additional and Prognostically Important Myocardial Infarctions in Patients Undergoing Percutaneous Coronary Intervention | Circulation: Cardiovascular Interventions

Abstract 16365: The Nature and Number of Unreported Events Identified by the Clinical Event Adjudication Process in the AEGIS-1 Trial | Circulation

7 Areas to Evaluate When Selecting Your Clinical Adjudication Partner - Paperpicks Leading Content Syndication and Distribution Platform